Faculty

Professor of Chemistry
Director of Graduate Admissions
Dr. Grabow received his PhD in Chemical Engineering from the University of Wisconsin in 2008, followed by postdoctoral appointments at the Technical University of Denmark and Stanford University. His expertise is the application of electronic structure calculations, kinetic modeling, data science and transient kinetic characterization to problems in heterogeneous catalysis, surface science and electrochemical energy storage. His papers have been highly cited and he was elected into the 2018 Class of Influential Researchers by Industrial and Engineering Chemistry (IE&C) Research. Prof. Grabow won the prestigious U.S. Department of Energy (DOE) Early Career Award (2014) and the NSF CAREER Award (2015), the Excellence in Research Award at the assistant professor level from the University of Houston (2017), among others. He currently serves as Chair of the Southwest Catalysis Society (SWCS), as Editor of Surface Science and on the International Advisory Board of ChemCatChem. He has previously served as (Vice/Past) Chair of the AIChE Catalysis and Reaction Engineering (CRE) Division and and is a past member of the Early Career Advisory Board of ACS Catalysis.
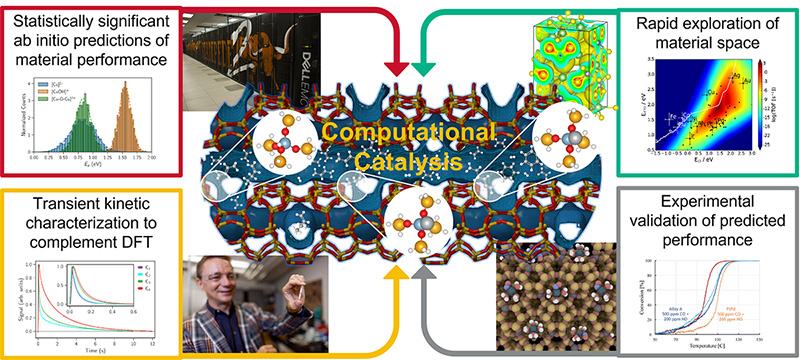
The Computational Catalysis and Interface Chemistry Group led by Prof. Grabow uses density functional theory, kinetic modeling, data science and transient kinetic characterization to provide molecular scale insight into fundamental questions related to reaction mechanisms and the nature of active sites in heterogeneous catalysis, surface science and electrochemical systems.
In addition to developing computational workflows to discover and design novel catalysts with tailored properties for chemical transformations relevant to the energy transition, the group explores the effect of externally imposed dynamics on catalytic systems to enhance catalytic performance. Transient kinetic experiments in a Temporal Analysis of Products (TAP) reactor are used for model validation and to develop dynamic processes. Given the multidisciplinary nature of the research project, the group collaborates extensively with researchers at UH and elsewhere, and is a member of the DOE Center for Programmable Energy Catalysis, The Center for Decarbonizing Chemical Manufacturing Using Sustainable Electrification (DC-MUSE), and The Center for Superconductivity at the University of Houston.
- Graduate students: Kai Wu (co-advised with Prof. Harold), Gary Gildert (co-advised with Prof. Harold), Dale Green, Kenneth Lucas Kusima, Md. Shariar Hossain, Fahmida Akter
-
Undergraduate students: Mikas Dunn
- Postdoctoral research associates: Shengguang Wang, Richard Tran, Hong Zhong
Andrea Prosperetti Research Computing Faculty Award, Cullen College of Engineering, University of Houston, 2021
Undergraduate Research Mentoring Award, University of Houston, 2020
First holder of the Dan Luss Endowed Professorship, 2019
Elected into the 2018 Class of Influential Researchers, Industrial & Engineering Chemistry (I&EC) Research, 2018
Best Fundamental Paper Award from the AIChE-STS (South Texas Section), 2017 & 2014
Excellence in Research, Scholarship or Creative Activity Award - Assistant Professor Level, University of Houston, 2017
Junior Faculty Research Excellence Award, Cullen College of Engineering, University of Houston, 2015
NSF CAREER Award, 2015
U.S. Department of Energy Early Career Award, 2014
Teaching Excellence Award, Cullen College of Engineering, University of Houston, 2014
Finalist in the Gerhard Ertl Young Investigator Award Competition, 2013
ICC Young Scientist Award, 2012
ACS-PRF Doctoral New Investigator Award, 2012
Graduation with Distinction Award from the University of Stuttgart, 2003
DAAD Scholarship (German Academic Exchange Service), 2001
AIChE Catalysis & Reaction Engineering Division: Programming Chair (2012 - 2014), 2nd Vice Chair, Vice Chair, Chair, Past Chair (2016 - 2020), Social Media Director (2019 - 2021)
Southwest Catalysis Society (SWCS): Director (2014 - 2016), Secretary (2017 - 2018), Vice Chair, Chair, Past Chair (2018 - 2022)
Editor of Surface Science, since 2020
International Advisory Board, (2017 - present)
Early Career Advisory Board of ACS Catalysis (2017 - 2018)
Review College of FWO (Fonds Wetenschappelijk Onderzoek Vlaanderen, The Research Foundation – Flanders)
Member of AIChE, ACS, NACS, SWCS, IPMI, AAAS, AVS.
Journal Papers / Refereed Journal Publications
- A. Mahdavi-Shakib, T. N. Whittaker, T. Yong Yun, K. B. S. Kumar, L. C. Rich, S. Wang, R. M. Rioux, L. C. Grabow, and B. D. Chandler, "The role of surface hydroxyls in the entropy-driven adsorption and spillover of H2 on Au/TiO2 catalyst", Nature Catalysis 6, 710-719 [DOI], 2023
- D. S. Mallapragada, Y. Dvorkin, M. Modestino, D. V. Esposito, W. Smith, B.-M. Hodge, M. P. Harold, V. M. Donnelly, A. Nuz, C. Bloomquist, K. Baker, L. C. Grabow, Y. Yan, N. N. Rajput, R. Hartman, E. J. Biddinger, E. Aydil, and A. Taylor, "Decarbonization of the Chemical Industry through Electrification: Barriers and Opportunities", Joule 7, 23–41 [DOI], 2023
- Y. Song, S. Svadlenak, T. Bathena, M. J. Hazlett, W. S. Epling, K. A. Goulas, and L. C. Grabow, "PdCu alloy catalyst for inhibition-free, low-temperature CO oxidation", ChemCatChem - accepted [DOI], 2023
- D. Parmar, S. Cha, T. Salavati-fard, A. Agarwal, H. Chiang, S. Washburn, J. Palmer, L. C. Grabow, J. D. Rimer, "Spatiotemporal Coke Coupling Enhances Para-Xylene Selectivity in Highly Stable MCM-22 Catalysts", Journal of the American Chemical Society 144, 7861-7870 [DOI], 2022
- T. M. Onn, S. Gathmann, S. Guo, S. P. Solanki, A. Walton, B. Page, G. Rojas, M. Neurock, L. C. Grabow, K. A. Mkhoyan, O. A. Abdelrahman, C. D. Frisbie, and P. Dauenhauer, "Platinum graphene catalytic condenser for millisecond programmable metal surfaces", Journal of the American Chemical Society 144, 48, 22113–22127 [DOI], 2022
- X. Li and L. C. Grabow, "Evaluating the Benefits of Kinetic Monte Carlo and Microkinetic Modeling for Catalyst Design Studies in the Presence of Lateral Interactions", Catalysis Today 387, 150-158 [DOI], 2022
- K. B. S. Kumar, T. Whittaker, C. Peterson, L. C. Grabow, B. D. Chandler, "Water Poisons H2 Activation at the Au-TiO2 Interface by Suppressing Electron Transfer between Au and Titania", Journal of the American Chemical Society 142, 5760-5772 [DOI], 2020
- K. Rao, Y. Yao, L. C. Grabow, "Accelerated Modeling of Lithium Diffusion in Solid State Electrolytes using Artificial Neural Networks", Advanced Theory and Simulations 3, 2000097 [DOI], 2020
- R. Nelson, B. Baek, P. Ruiz, B. Goundie, A. Brooks, M. C. Wheeler, B. G. Frederick, L. C. Grabow, and R. Narehood Austin, "Experimental And Theoretical Insights Into The Hydrogen-Efficient Direct Hydrodeoxygenation Mechanism Of Phenol Over Ru/TiO2", ACS Catalysis 5, 6509–6523 [DOI], 2015
- J. Saavedra, H. A. Doan, C. J. Pursell, L. C. Grabow*, B. D. Chandler*, "The critical role of water at the gold-titania interface in catalytic CO oxidation", Science 345, 1599-1602 [DOI], 2014
Link to file
Books
- Lars C. Grabow, “Computational Catalyst Screening” in “Computational Catalysis” edited by A. Asthagiri and M. J. Janik. RSC Catalysis Series, Cambridge, UK [DOI], 2014
JOURNAL PAPERS / REFEREED JOURNAL PUBLICATIONS
- L. C. Grabow, J. J. Uhlrich, T. F. Kuech, M. Mavrikakis, “Effectiveness of in-situ NH3 annealing treatments for the removal of oxygen from GaN(0001) surfaces” Surf. Sci. 603, 387-399 [DOI], 2009
- L. R. Merte, J. Knudsen, L. C. Grabow, R. T. Vang, E. Lægsgaard, M. Mavrikakis, F. Besenbacher, “Correlating STM contrast and atomic-scale structure by chemical modification: Vacancy dislocation loops on FeO/Pt(111)”, Surf. Sci. 603, L15-L18 [DOI], 2009
- J.J. Uhlrich, L. C. Grabow, M. Mavrikakis, T. F. Kuech, “Practical Surface Treatments and Surface Chemistry of n-Type and p-Type GaN”, J. Elec. Mat. 37, 439 [DOI], 2008
- L. C. Grabow, A. A. Gokhale, S. Evans, J. A. Dumesic, M. Mavrikakis, “Mechanism of the water gas shift reaction on Pt: First principles, experiments, and microkinetic modeling”, J. Phys. Chem. C 112, 4608 [DOI], 2008
- L. C. Grabow, M. Mavrikakis, “Nanocatalysis Beyond the Gold-Rush Era”, Angew. Chem. Int. Ed. 47, 7390-7392 [DOI], Angew. Chem. 120, 7500-7502 [DOI], 2008
- N. Schumacher, K. Andersson, L. C. Grabow, M. Mavrikakis, J. Nerlov, I. Chorkendorff, “Interaction of carbon dioxide with Cu overlayers on Pt(111)”, Surf. Sci. 602, 702 [DOI], 2008
- S. Seo, L. C. Grabow, M. Mavrikakis, R. J. Hamers, N. J. Thompson, P. Evans, “Molecular-scale structural distortions near vacancies in pentacene”, Appl. Phys. Lett. 92, 153313 [DOI], 2008
- L. C. Grabow, Y. Xu, M. Mavrikakis, “Lattice strain effects on the CO oxidation on Pt(111)”,Phys. Chem. Chem. Phys. 8, 3369-3374 [DOI] (featured as cover page image), 2006
- N. Schumacher, A. Boisen, S. Dahl, A. A. Gokhale, S. Kandoi, L. C. Grabow, J. A. Dumesic, M. Mavrikakis, I. Chorkendorff, “Trends in low temperature water-gas shift reactivity on transition metals”, J. Cat. 229, 265 [DOI], 2005
- S. Kandoi, A. A. Gokhale, L. C. Grabow, J. A. Dumesic, M. Mavrikakis, “Why Au and Cu Are More Selective Than Pt for Preferential Oxidation of CO at Low Temperature”, Catal. Lett.93, 93 [DOI], 2004